Publications & Presentations
Publications
Articles about the YODA Project
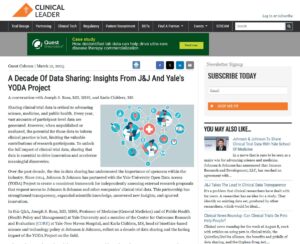
Ross JS, Childers K. A Decade Of Data Sharing: Insights From J&J And Yale’s YODA Project. Clinical Leader. 2025.

Vazquez E, Gouraud H, Naudet F, Gross CP, Krumholz HM, Ross JS, Wallach JD. Characteristics of available studies and dissemination of research using major clinical data sharing platforms. Clin Trials. 2021 Aug 18:17407745211038524. doi: 10.1177/17407745211038524

Ross JS, Waldstreicher J, Krumholz HM. Sharing clinical trial data: lessons from the YODA Project. STAT News. 2019.

Ross JS, Waldstreicher J, Bamford S, Berlin JA, Childers K, Desai NR, Gamble G, Gross CP, Kuntz R, Lehman R, Lins P, Morris SA, Ritchie JD, Krumholz HM. Overview and experience of the YODA Project with clinical trial data sharing after 5 years. Sci Data. 2018;5:180268.

Wilcox MA, Savitz AJ, Addington AM, Gray GS, Guinan EC, Jackson JW, Lehner T, Normand SL, Ranu H, Senthil G, Spertus J, Valeri L, Ross JS. The Open Translational Science in Schizophrenia (OPTICS) project: an open-science project bringing together Janssen clinical trial and NIMH data. NPJ Schizophr. 2018;4(1):14.

Low J, Ross JS, Ritchie JD, Gross CP, Lehman R, Lin H, Fu R, Stewart LA, Krumholz HM. Comparison of two independent systematic reviews of trials of recombinant human bone morphogenetic protein-2 (rhBMP-2): the Yale Open Data Access Medtronic Project. Systematic Reviews. 2017;6:28.

Krumholz HM, Waldstreicher J. The Yale Open Data Access (YODA) Project- A mechanism for data sharing. NEJM. 2016;375:403-405.

Ross JS, Addington AM, Senthil G, Lehner T, Wilcox MA. The OPTICS Project: An open-science framework for the analysis of clinical trial data. Psychiatric Times. 2016;33.

Krumholz HM, Ross JS, Gross CP, Emanuel EJ, Hodshon B, Ritchie JD, Low JB, Lehman R. A Historic moment for open science: The Yale University Open Data Access Project and Medtronic. Ann Intern Med. 2013;158:910-911.

Krumholz HM, Ross JS. A model for dissemination and independent analysis of industry data. JAMA. 2011;306:1593-1594.
Articles about Data Sharing

Vazquez E, Ross JS, Gross CP, Childers K, Bamford S, Ritchie JD, Waldstreicher J, Krumholz HM, Wallach JD. Concordance between clinical trial data use request proposals and corresponding publications: A cross-sectional study. Clinical Trials. 2025;0(0). doi:10.1177/17407745241304355

Ross JS, Waldstreicher J, Krumholz HM. Data Sharing – A New Era for Research Funded by the U.S. Government. N Engl J Med. 2023 Nov 15. doi: 10.1056/NEJMp2308792. Epub ahead of print. PMID: 37966303.

Rowhani-Farid A, Grewal M, Solar S, Eghari A, Zhang A, Gross C, Krumholz H, Ross J. Clinical trial data sharing: a cross-sectional study of outcomes associated with two U.S. National Institutes of Health models. Sci Data. 2023:10;529. https://doi.org/10.1038/s41597-023-02436-0

Tannenbaum S, Ross JS, Krumholz HM, Desai NR, Ritchie JD, Lehman R, Gamble GM, Bachand J, Schroter S, Groves T, Gross CP. Early experiences with journal data sharing policies: A survey of published clinical trial investigators. Ann Intern Med. [Epub ahead of print 17 July 2018] doi: 10.7326/M18-0723.

Dey P, Ross JS, Ritchie JD, Desai NR, Bhavnani SP, Krumholz HM. Data sharing and cardiology: Platforms and possibilities. JACC. 2017;70:3018-3025.

Angraal S, Ross JS, Desai N, Dhruva SS, Krumholz HM. Research Letter: Merits of data sharing: the Digitalis Investigation Group trial. JACC. 2017;70:1825-1826.

Ross JS, Ritchie JD, Finn E, Desai NR, Lehman RL, Krumholz HM, Gross CP. Data sharing through an NIH central database repository: A cross-sectional survey of BioLINCC users. BMJ Open. 2016;6:e012769.

Ross JS. Clinical research data sharing: What an open science world means for researchers involved in evidence synthesis. Systematic Reviews. 2016;5:159.

Krumholz HM, Bourne PE, Kuntz RE, Paz HL, Terry SF, Waldstreicher J. Data acquisition, curation, and use for a continuously learning health system: A vital direction for health and health care. JAMA. 2016;epub ahead of print.

Murugiah K, Ritchie JD, Desai NR, Ross JS, Krumholz HM. Availability of clinical trial data from industry-sponsored cardiovascular trials. J Am Heart Assoc. 2016;5:e003307.

Krumholz HM. Why data sharing should be the expected norm. BMJ. 2015;350:h599.

Rathi VK, Strait KM, Gross CP, Hrynaszkiewicz I, Joffe S, Krumholz HM, Dzara K, Ross JS. Predictors of clinical trial data sharing: Exploratory analysis of a cross-sectional survey. Trials. 2014;15:384.

Krumholz HM, Peterson ED. Open access to clinical trials data. JAMA. 2014;312:1002-1003.

Krumholz HM, Gross CP, Blount KL, Ritchie JD, Hodshon B, Lehman R, Ross JS. Sea change in open science and data sharing: leadership by industry. Circ Cardiovasc Qual Outcomes. 2014;7:499-504.

Ross JS, Krumholz HM. Ushering in a new era of open science through data sharing: The wall must come down. JAMA. 2013;309:1355-1356.

Ross JS, Krumholz HM. Implications of clinical trial data sharing for medical writers. Medical Writing. 2013;22:45-49.

Rathi V, Dzara K, Gross CP, Hrynaszkiewicz I, Joffe S, Krumholz HM, Strait KM, Ross JS. Sharing of clinical trial data among trialists: A cross sectional survey. BMJ. 2012;345:e7570.

Krumholz HM. Open science and data sharing in clinical research: Basing informed decisions on the totality of the evidence. Circ Cardiovasc Qual Outcomes. 2012;5:141-142.

Ross JS, Lehman R, Gross CP. The importance of clinical trial data sharing: Toward more open science. Circ Cardiovasc Qual Outcomes. 2012;5:238-240.

Krumholz HM. Independent analysis of industry data necessary for promoting collaborative science. Cardiology. 2012;41:9.

Lehman R, Loder E. Missing clinical trial data. BMJ. 2012;344:d8158.
Articles about Transparency

Zou CX, Becker JE, Phillips AT, Garritano JM, Krumholz HM, Miller JE, Ross JS. Registration, results reporting, and publication bias of clinical trials supporting FDA approval of neuropsychiatric drugs before and after FDAAA: a retrospective cohort study. Trials. 2018;19(1):581.

Phillips AT, Desai NR, Krumholz HM, Zou CX, Miller JE, Ross JS. Association of the FDA Amendment Act with trial registration, publication, and outcome reporting. Trials. 2017;18:333.

Krumholz HM, Kim J. Data escrow and clinical trial transparency. Ann Intern Med. [Epub ahead of print 4 April 2017] doi: 10.7326/M17-0743

Phillips AT, Rathi VK, Ross JS. Publication of clinical studies supporting FDA premarket approval for high-risk cardiovascular devices between 2011 and 2013: A cross-sectional study. JAMA Intern Med. 2016;176:551-552.

Chen R, Desai NR, Ross JS, Zhang W, Chau KH, Wayda B, Murugiah K, Lu DY, Mittal A, Krumholz HM. Publication and reporting of clinical trial results: Cross sectional analysis across academic medical centers. BMJ. 2016;352:i637.

Gopal AD, Desai NR, Tse T, Ross JS. Reporting of noninferiority trials in ClinicalTrials.gov and corresponding publications. JAMA. 2015;313:1163-1165.

Ross JS. Confronting bias. JAMA Intern Med. 2015;175:307.

Lauer MS, Krumholz HM, Topol EJ. Time for a prepublication culture in clinical research? Lancet. 2015;386:2447-2449.

Shaw DL, Ross JS. US Federal Government efforts to improve clinical trial transparency with expanded trial registries and open data sharing. AMA Journal of Ethics. 2015;17:1152-1159.

Krumholz HM. The end of journals. Circ Cardiovasc Qual Outcomes. 2015;8:00-00.
Smithy JW, Downing NS, Ross JS. Publication of pivotal efficacy trials for novel therapeutic agents approved between 2005 and 2011: A cross-sectional study. JAMA Intern Med. 2014;174:1518-1520.

Lehman R. The medical reformation. BMJ. 2014;348:g3963.
Becker JE, Krumholz HM, Ben-Josef G, Ross JS. Reporting of results in ClinicalTrials.gov and high-impact journals. JAMA. 2014;311:1063-1065.

Ross JS, Mocanu M, Lampropulos JF, Tse T, Krumholz HM. Time to publication among completed clinical trials. JAMA Intern Med. 2013;173:825-828.

Ross JS, Gross CP, Krumholz HM. Promoting transparency in pharmaceutical industry–sponsored research. Am J Public Health. 2012;102:72-80.

Ross JS, Tse T, Zarin DA, Xu H, Zhou L, Krumholz HM. Publication of NIH funded trials registered in ClinicalTrials.gov: Cross sectional analysis. BMJ. 2011;344:d7292.

Ross JS, Mulvey GK, Hines EM, Nissen SE, Krumholz HM. Trial publication after registration in ClinicalTrials.Gov: A cross-sectional analysis. PLoS Med. 2009;6:e1000144.

Krumholz HM. Registries and selection bias: The need for accountability. Circ Cardiovasc Qual Outcomes. 2009;2:517-518.
Other

Wallach JD, Wang K, Zhang AD, Cheng D, Grossetta Nardini HK, Lin H, Bracken MB, Desai M, Krumholz HM, Ross JS. Updating insights into rosiglitazone and cardiovascular risk through shared data: individual patient and summary level meta-analyses. BMJ. 2020 Feb 5;368:l7078.
Approved research proposal through ClinicalStudyDataRequest.com. You can view the results of the study on the MedRxiv website.

Krumholz HM. How Do We Know the Value of Our Research? Circ Cardiovasc Qual Outcomes. 2013;6:371-372.
Presentations

“Sharing Patient-Level Clinical Trial Data: The YODA Model” (Given by Cary P. Gross, MD at the National Brain Tumor Society Research Roundtable, July 20, 2023)

“The YODA Project: A Use Case and Lessons Learned” (Given by Joseph S. Ross, MD, MHS, at the National Academies Challenges and a Way Forward in Sharing Clinical Trial Data Workshop, November 18, 2019)

“Sharing Data through the YODA Project: Early Experiences and Lessons Learned” (Given by Joseph S. Ross, MD, MHS, at the Center for Biomedical Research Transparency Summit, February 15, 2019)

“Openness About Clinical Trial Results: Lessons from the Front Line” (Given by Joseph S. Ross, MD, MHS, at the American Association for the Advancement of Science Annual Meeting, February 15, 2019)

“Sharing Data through the YODA Project: Early Experience” (Given by Joseph S. Ross, MD, MHS, at the Eighth International Congress on Peer Review and Scientific Publication, September 10, 2017)

“Clinical Trial Data Sharing and Reproducibility” (Given by Harlan M. Krumholz, MD, SM, and Jeffrey M. Drazen, MD, at the Harvard Medical School Health Policy and Bioethics Consortium Meeting, December 7, 2016)

“The Yale Open Data Access (YODA) Project: Repurposing Data for Researchers” (Given by Jessica Ritchie Klein, MPH, PMP, at the American Library Association Annual Meeting, June 27, 2016)

“The Yale Open Data Access (YODA) Project: Lessons Learned in Data Sharing” (Given by Joseph S. Ross, MD, MHS, at the Society for Clinical Trials Annual Meeting, May 18, 2016)

“Data Sharing: Perspectives from a Third Party, Independent Academic Process (YODA)” (Given by Joseph S. Ross, MD, MHS, at the CBI Conference on Clinical Data Disclosure and Transparency, January 29, 2015)

“Clinical Trial Data Sharing” (Given by Joseph S. Ross, MD, MHS, at the NPA FDA Taskforce Meeting, December 15, 2014)

“BMP Use: Evaluating Industry-Funded Trials” (Given by Kevin Bozic, MD, at the American Academy of Orthopaedic Surgeons (AAOS), Orthopaedic Research Society (ORS) Clinical Research Forum, February 6, 2012)

“A Model for Dissemination and Independent Analysis of Clinical Trial Program Data” (Given by Harlan Krumholz, MD, at the American College of Cardiology (ACC) Board of Trustees Meeting, December 5, 2011)
